Research
1.Vaccines
2.Infectious Diseases
3.Immunity
1. Vaccines
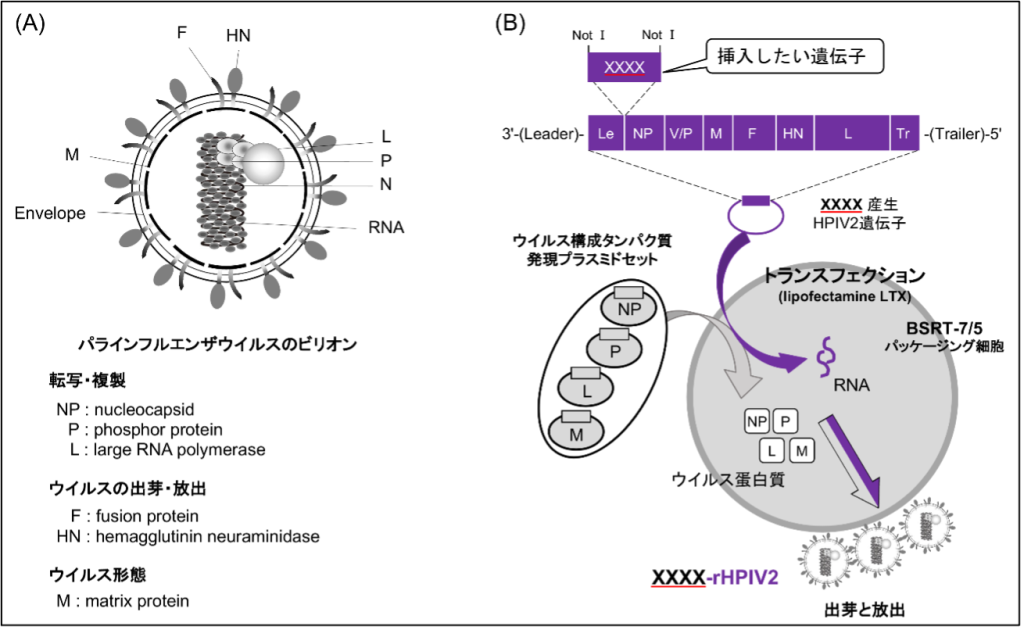
1. Nasal Vaccine
Using a human para-influenza virus 2 (HPIV2) for a respiratory tract infection in humans, we are investigating a highly safe nasal vaccine that targets the respiratory mucosa. Currently, we are producing vaccines with tuberculosis and AIDS genes inserted into them.
2. Recombinant BCG Vaccine
Tuberculosis was the leading cause of death from a single infectious disease prior to the COVID-19 pandemic. The only licensed TB vaccine, BCG, is administered at approximately 6 months of age and protect them from tuberculosis meningitis and miliary tuberculosis up to 80%. However, because the BCG vaccine is a live attenuated vaccine, it is prohibited in immunocompromised individuals, e.g. HIV-positive patients and those receiving immunosuppressive treatment. Therefore, there is a need for the development of a more effective BCG vaccine against tuberculosis that has fewer side effects.
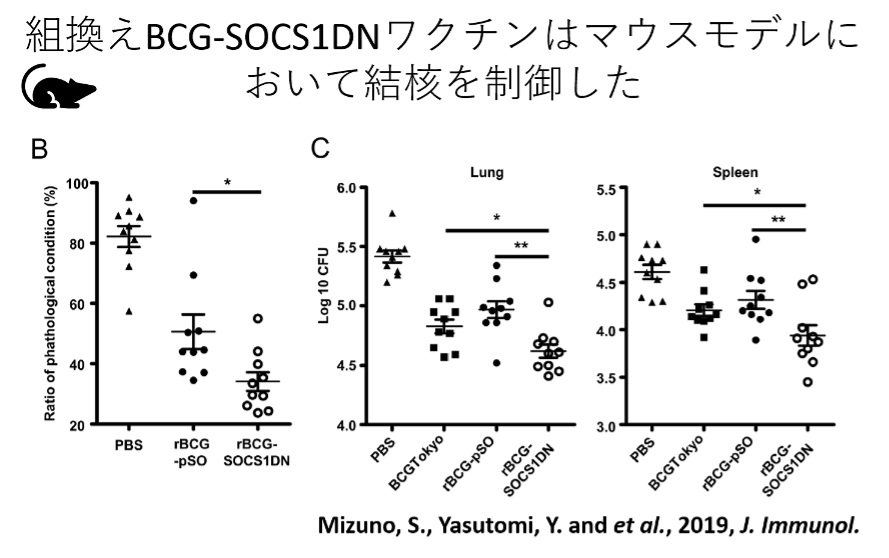
Suppressor of cytokine signaling 1 (SOCS1) is a molecule that negatively regulates JAK/STAT signaling in immune cells. A recombinant BCG-SOCS1DN vaccine with a mutation in the active site of SOCS1 was produced and inoculated into an immunodeficient mouse model (RAG1 knockout mice). They showed no pathogenic response [Soma, S. et al., Infect. Immun., 86, e00376-18, 2018]. Furthermore, when this vaccine was administered into wild-type mice, it showed a more protective effect against tuberculosis than the existing BCG vaccine (BCG Tokyo strain) [Mizuno, S., et al., J. Immunol., 203, pp188-197, 2019]. In our study using cynomolgus macaques, the recombinant BCG-SOCS1DN vaccine was as effective as the BCG Tokyo strain in protecting against tuberculosis in healthy subjects. Pathogenicity was not observed in healthy subjects, thus proving to be a promising novel BCG vaccine.
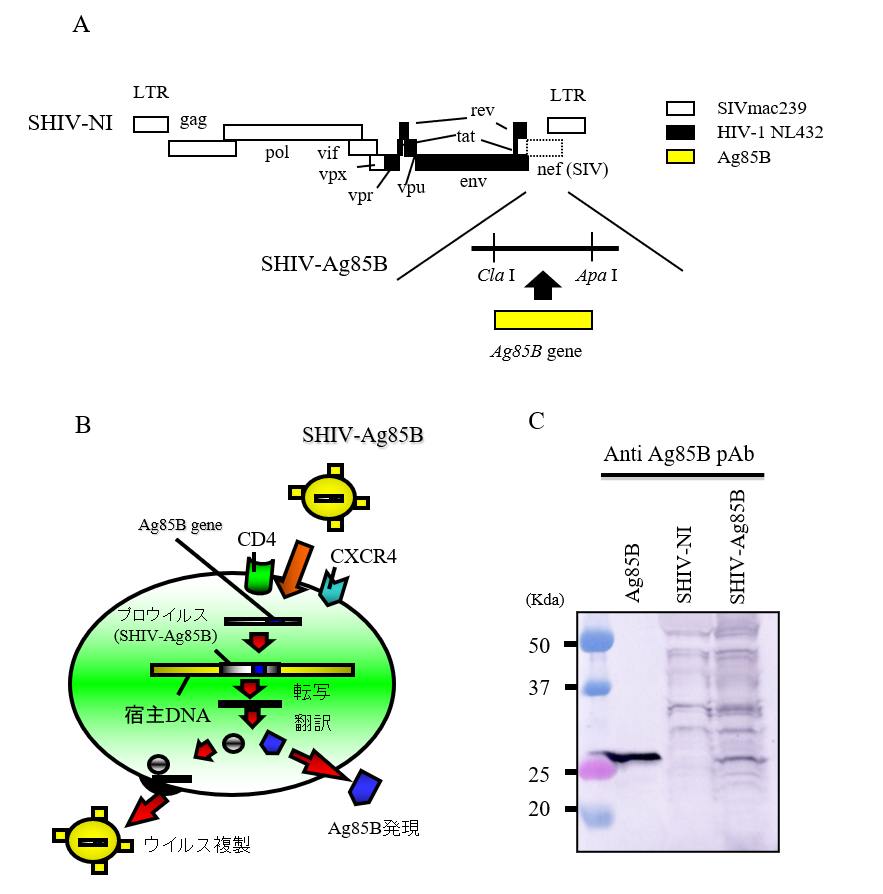
Ag85B-adjuvanted Attenuated AIDS Vaccine
We constructed an attenuated AIDS vaccine by inserting Ag85B as an adjuvant into a Nef-deficient attenuated AIDS vaccine. This vaccine’s immune response and its effectiveness is currently under investigation in cynomolgus monkeys.
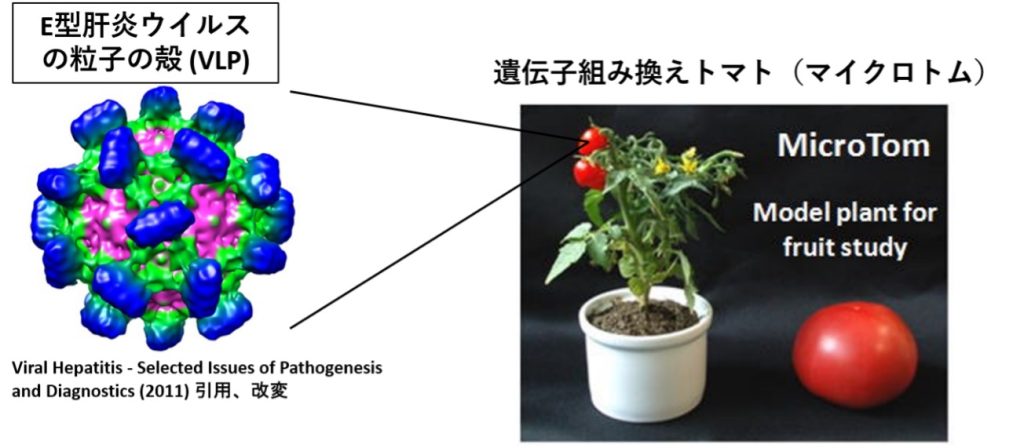
Oral Vaccine (Tomato)
We are utilizing orally transmitting Hepatitis E Virus (HEV) Virus Like-Particles (VLPs) for an oral vaccine. On the surface of the VLPs, we expressed other viral antigens in a chimera VLP vaccine. Alternatively, we inserted a plasmid expressing viral antigen into the VLPs. Currently, in our collaboration with the University of Tsukuba, we are producing chimeric VLP-expressing tomatoes for an edible vaccine. The vaccine’s effects against HEV and influenza are currently under investigation.
Infectious Diseases
Novel Coronavirus
We are establishing a Novel Coronavirus infection model using cynomolgus monkeys and transgenic mice. In our infection model using CAG promoter-hACE2 mice, the mice were highly sensitive to SARS-CoV-2 and expressed pathogenicity against various strains. Moreover, administration of a neutralizing antibody or an anti-viral drug led to improvements in decreasing body weight rate and decreasing survival rate. Currently, we are continuing this investigation on the effects of the neutralizing antibody and the anti-viral drug in our infection models.
Monkeypox
Monkeypox is caused by the monkeypox virus leading to an acute rash. Its natural host is suspected to be rodents inhabiting in Africa. Under the Infectious Diseases Prevention Law, monkeypox is a Category 4 virus. Here at Tsukuba Primate Research Center, we are currently investigating the prevention and treatment of monkeypox using cynomolgus monkeys.
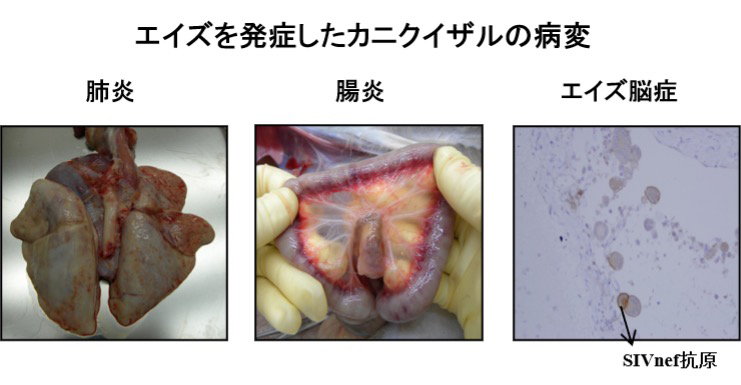
HIV
We infect macaques including cynomolgus monkeys with SIV or SHIV as an AIDS animal model. Using this model, we are evaluating new AIDS vaccines and anti-HIV therapeutic treatment.
HBV
HBV has high host specificity, where except for the tree shrew, infection of HBV is only established in apes such as humans and chimpanzees. As a result of the prohibition of animal experiments on chimpanzees by animal welfare, a large amount of HBV experiments using animals involve chimeric mice with human livers. However, due to the loss of immune function in these animals, research on therapeutic methods for HBV is proven to be difficult in application. Therefore, we are managing methods of raising, breeding, and nurturing pilot colonies of 100 tree shrews. We aim to establish a highly HBV-infective tree shrew.
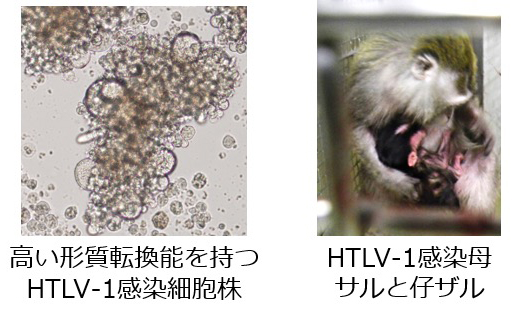
HTLV-1
Using an in vitro HTLV-1 cell line that has high efficiency transformation, we have successfully infected cynomolgus monkeys. Using this model, we aim to elucidate the pathology of HTLV-1 by analyzing immune function during infection. Furthermore, we are investigating the development of preventative medicine and vaccines for HTLV-1. We are also continuing our investigation on vertical infection monkey model.
Immunity
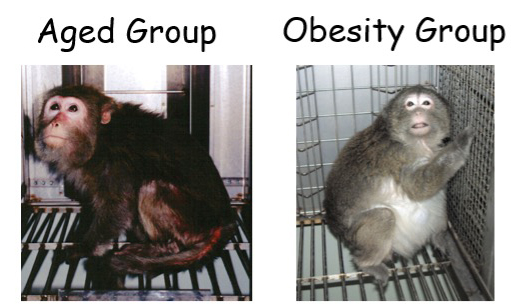
Aged Monkeys, Obese Monkeys
We are the only primate research center worldwide who houses aged monkeys. We also found that obesity in some of our monkeys leads to metabolic disorders. Therefore, using colonies of cynomolgus monkeys, we are investigating the effects of aging and obesity on immunity. In our investigations, we are mainly analyzing our results in regards to infection and on reduced immune function.
Lipidomics
In recent research, large changes in metabolites found in the blood due to infection are becoming well-known. We try to perform a metabolomic analysis using plasma from monkeys infected with tuberculosis to identify factors corelated with vaccine effects and find new therapeutic drug targets.
